UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
CURRENT REPORT
PURSUANT TO SECTION 13 OR
15(d) of the SECURITIES EXCHANGE ACT
OF 1934
Date of Report (Date of earliest event reported): August 13, 2019
VistaGen Therapeutics, Inc.
(Exact name of registrant as specified in its charter)
|
NEVADA
|
001-37761
|
20-5093315
|
|
(State or other jurisdiction of incorporation)
|
(Commission File Number)
|
(IRS Employer Identification Number)
|
|
343 Allerton Ave.
South San Francisco, California 94090
|
|
(Address of principal executive offices)
|
(650) 577-3600
(Registrant’s telephone number, including area
code)
Not Applicable
(Former name or former address, if changed since last
report)
Check the appropriate box below if the Form 8-K filing is intended
to simultaneously satisfy the filing obligation of the registrant
under any of the following provisions:
☐ Written communications
pursuant to Rule 425 under the Securities Act (17 CFR
230.425)
☐ Soliciting material
pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a
-12)
☐ Pre-commencement
communications pursuant to Rule 14d-2(b) under the Exchange Act (17
CFR 240.14d -2(b))
☐ Pre-commencement
communications pursuant to Rule 13e-4(c) under the Exchange Act (17
CFR 240.13e -4(c))
Securities registered pursuant to Section 12(b) of the
Act:
|
Title of each class
|
Trading Symbol(s)
|
Name of each exchange on which
registered
|
|
Common
Stock, par value $0.001 per share
|
VTGN
|
Nasdaq
Capital Market
|
Indicate by check mark whether the registrant is
an emerging growth company as defined in Rule 405 of the Securities
Act of 1933 (17 CFR 230.405) or Rule 12b-2 of the Securities
Exchange Act of 1934 (17 CFR 240.12b-2) ☐
If an emerging growth company, indicate by check
mark if the registrant has elected not to use the extended
transition period for complying with any new or revised financial
accounting standards provided pursuant to Section 13(a) of the
Exchange Act ☐
Item 2.02 Results of Operations and Financial
Condition.
On August 13, 2019, VistaGen Therapeutics, Inc.
(the “Company”) issued a press release to announce the
Company’s financial results for its fiscal year 2020 first
quarter ended June 30, 2019. A copy of the press release is
attached to this Current Report on Form 8-K as Exhibit
99.1.
Item 7.01 Regulation FD Disclosure.
See
Item 8.01.
Item 8.01 Other Items.
On
August 15, 2019, the Company announced that it has achieved
completion of target patient enrollment of 180 patients in the
Company’s Phase 2 ELEVATE clinical trial. ELEVATE is a
multi-center, double-blind, placebo-controlled clinical study to
evaluate the efficacy and safety of AV-101, the Company’s
novel, oral NMDA (N-methyl-D-aspartate) receptor glycine site
antagonist, as an adjunctive treatment (together with an
FDA-approved oral antidepressant) for major depressive disorder in
adult patients with an inadequate therapeutic response to their
current antidepressants. The Company expects to report topline
results from the ELEVATE study before the year end 2019. A copy of
the press release is attached to this Current Report on Form 8-K as
Exhibit 99.2.
On
August 15, 2019, the Company began utilizing a new corporate
presentation. A copy of the updated corporate presentation is
attached to this Current Report on Form 8-K as Exhibit
99.3.
The information in this Current Report
on Form 8-K, including the information set forth in
Exhibits 99.1 and 99.3, is being furnished and shall not be
deemed “filed” for purposes of Section 18 of the
Securities Exchange Act of 1934, as amended (the
“Exchange
Act”), nor shall Exhibits
99.1 and 99.3 filed herewith be deemed incorporated by reference in
any filing under the Securities Act of 1933, as amended, or the
Exchange Act, except as shall be expressly set forth by specific
reference in such a filing.
Item 9.01 Financial Statements and Exhibits.
(d) Exhibits Index
|
Exhibit No.
|
|
Description
|
|
|
|
|
|
|
Press
Release issued by VistaGen Therapeutics, Inc., dated August 13,
2019.
|
|
|
|
Press
Release issued by VistaGen Therapeutics, Inc., dated August 15,
2019.
|
|
|
|
VistaGen
Therapeutics, Inc. Corporate Presentation, dated August
2019.
|
Signatures
Pursuant to the
requirements of the Securities Exchange Act of 1934, the registrant
has duly caused this report to be signed on its behalf by the
undersigned thereunto duly authorized.
|
|
VistaGen
Therapeutics, Inc.
|
|
|
|
|
|
|
Date:
August 16, 2019
|
By:
|
/s/ Shawn K.
Singh
|
|
|
|
Shawn
K. Singh
Chief
Executive Officer
|
EXHIBIT INDEX
|
Exhibit No.
|
|
Description
|
|
|
|
|
|
|
Press
Release issued by VistaGen Therapeutics, Inc., dated August 13,
2019.
|
|
|
|
Press
Release issued by VistaGen Therapeutics, Inc., dated August 15,
2019.
|
|
|
|
VistaGen
Therapeutics, Inc. Corporate Presentation, dated August
2019.
|
Exhibit 99.1

VistaGen Therapeutics Reports Fiscal 2020 First Quarter Financial
Results
SOUTH SAN FRANCISCO, Calif., August 13, 2019 - VistaGen
Therapeutics (NASDAQ: VTGN), a clinical-stage
biopharmaceutical company developing new generation medicines for
central nervous system (CNS) diseases and disorders with high unmet
need, today announced financial results for its fiscal year 2020
first quarter ended June 30, 2019.
“The next few months are potentially transformative for
VistaGen, as we look forward to several clinical and regulatory
milestones before year-end,” stated Shawn
Singh, Chief Executive Officer of VistaGen. “We have three
differentiated clinical-stage drug candidates, each of which has an
exceptional safety profile in studies to date and significant
therapeutic and commercial potential in multiple large and growing
CNS markets where current treatments are inadequate to meet the
needs of millions of patients. Our team is focused on driving
continued progress across our pipeline, and we are confident in our
efforts to achieve our core goals – to deliver both safe and
effective new generation treatments in neuropsychiatry and
neurology for patients and extraordinary value to our loyal
shareholders.”
Financial Results for the Fiscal Quarter Ended June 30,
2019:
Net loss attributable to common stockholders for the fiscal quarter
ended June 30, 2019 was approximately $6.2 million, including
approximately $1.2 million of noncash charges, compared to $4.2
million for the fiscal quarter ended June 30, 2018, primarily
attributable to increased research and development activities
relating to the Company’s CNS drug development
programs.
Research and development expense totaled $4.3 million for the
fiscal quarter ended June 30, 2019, compared with $2.7 million for
the fiscal quarter ended June 30, 2018. The increase in research
and development expense is primarily related to the continued
progress of ELEVATE, the Company’s Phase 2 clinical study
evaluating efficacy and safety of AV-101, its novel oral NMDA
(N-methyl-D-aspartate) receptor glycine site antagonist, as an
add-on treatment (together with an FDA-approved oral
antidepressant) for adults with major depressive disorder (MDD),
several preclinical studies, including studies supporting
AV-101’s potential for treating neuropathic pain (NP) and
levodopa-induced dyskinesia (LID) in patients with
Parkinson’s disease, and manufacturing activities involving
AV-101 and the Company’s two novel, clinical-stage
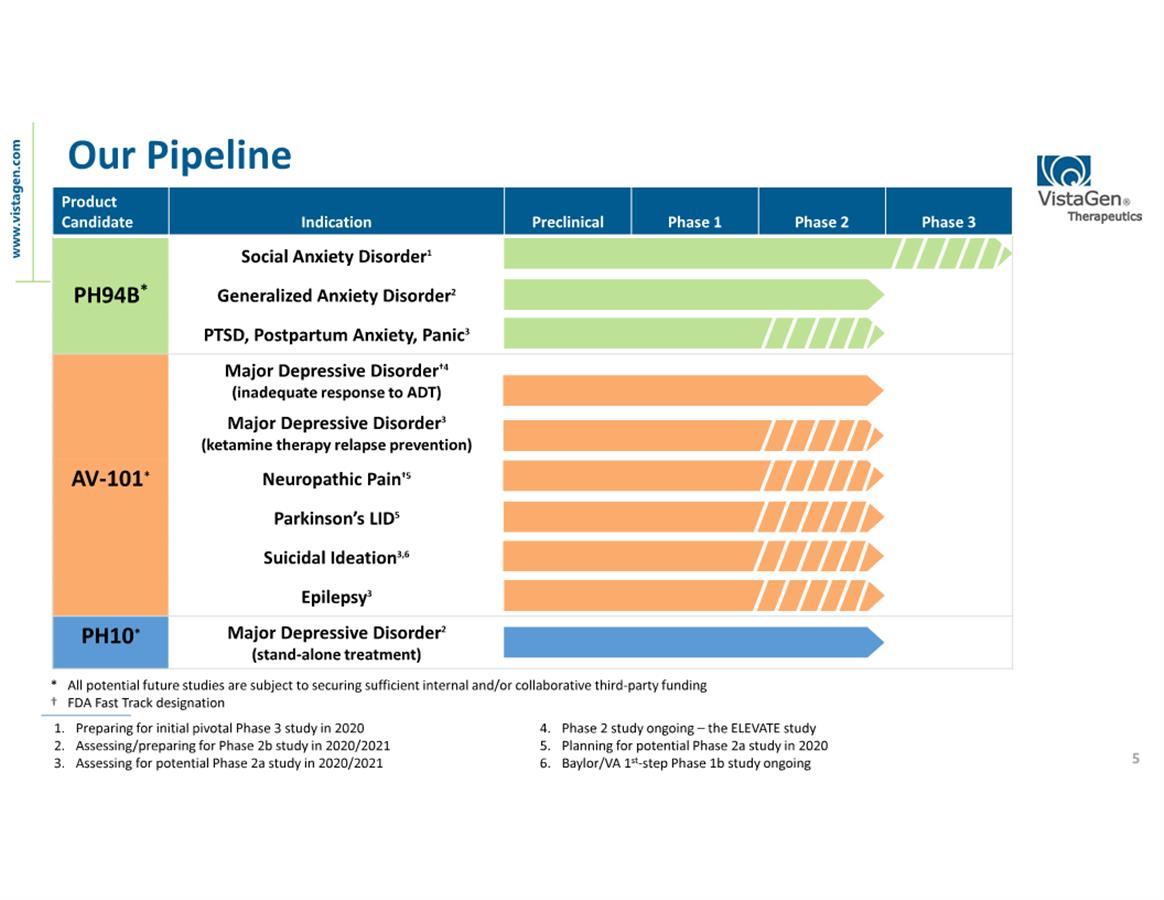
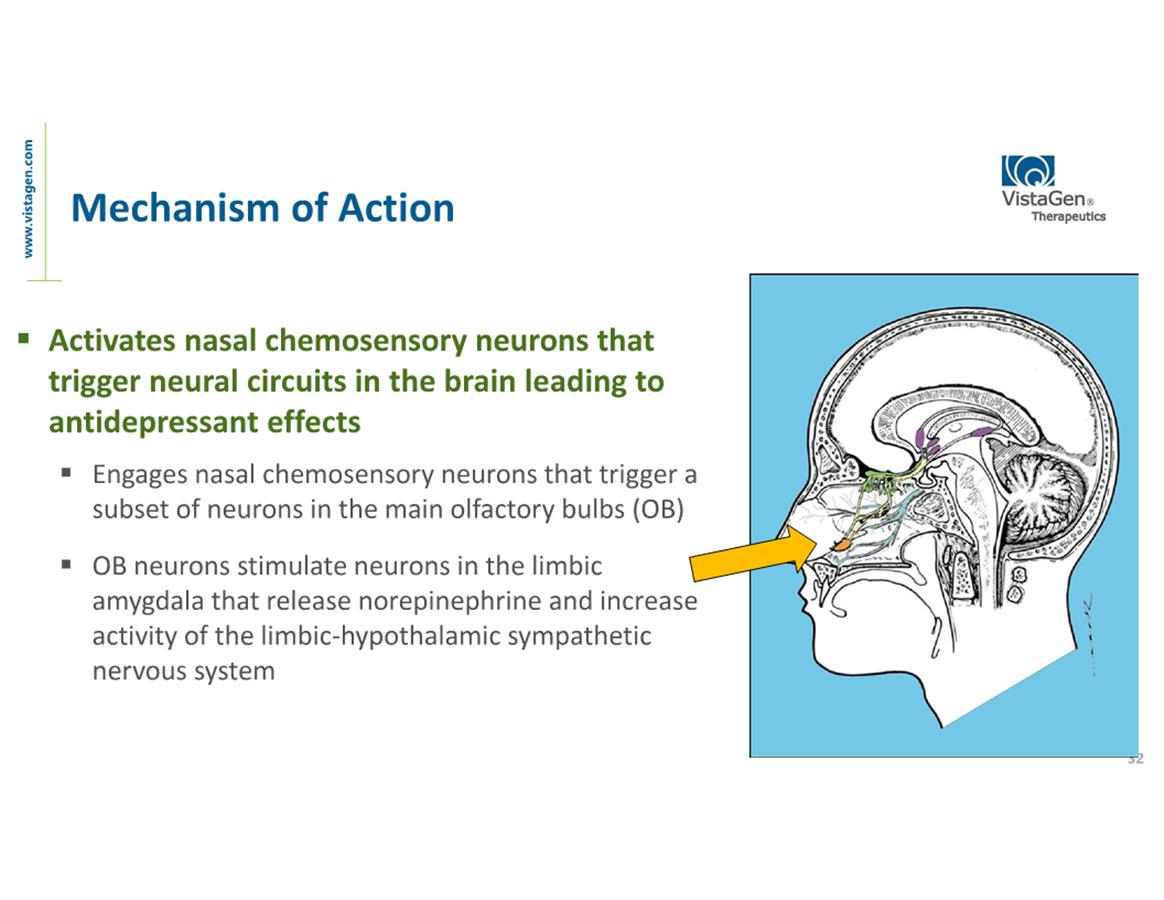
neuroactive nasal spray candidates, PH94B for social anxiety
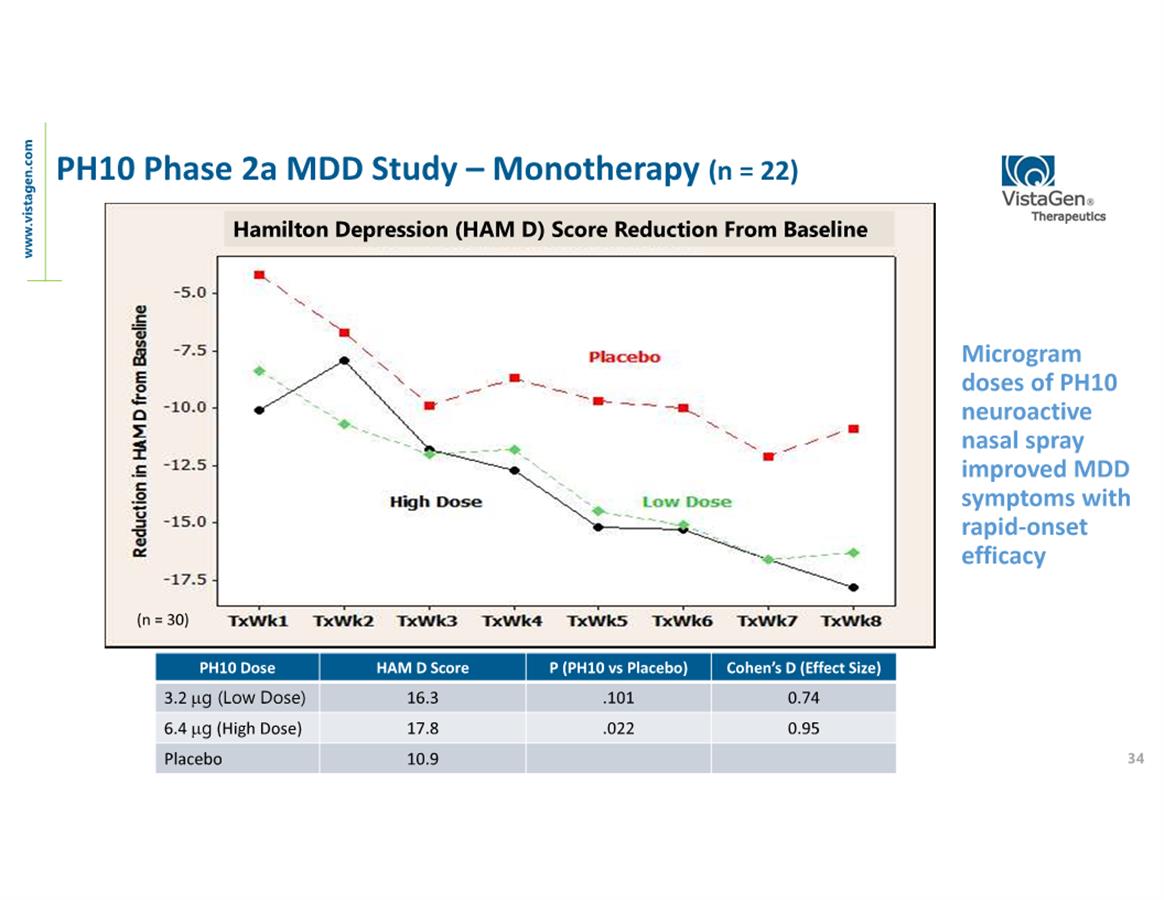
disorder (SAD) and PH10 for MDD.
General and administrative expense was approximately $1.9 million
in the fiscal quarter ended June 30, 2019, compared to
approximately $1.5 million in the fiscal quarter ended June 30,
2018. The increase was primarily attributable to noncash stock
compensation expense.
At June 30, 2019, VistaGen had cash and cash equivalents of $8.3
million, compared to $13.1 million at March 31, 2019.
As of August 13, 2019, there were 42,622,965 shares of common stock
outstanding.
-1-
About VistaGen
VistaGen Therapeutics is a clinical-stage biopharmaceutical company
developing new generation medicines for CNS diseases and disorders
where current treatments are inadequate, resulting in high unmet
need. VistaGen’s
pipeline includes
three differentiated, clinical-stage CNS drug candidates, AV-101,
PH10 and PH94B, each with an exceptional safety profile in all
clinical studies to date and therapeutic potential in multiple
large and growing CNS markets. For more information, please
visit www.vistagen.com and
connect with VistaGen on Twitter, LinkedIn and Facebook.
Forward-Looking Statements
This release contains various statements concerning VistaGen's
future expectations, plans and prospects, including without
limitation, our expectations regarding development and
commercialization of our three drug candidates: (i) AV-101 for MDD,
NP, LID and suicidal ideation; (ii) PH94B for SAD; and (iii) PH10
for MDD. In addition, statements concerning the Company’s
future expectations may include statements regarding intellectual
property and commercial protection of our drug candidates. Each of
these statements constitute forward-looking statements for the
purposes of the safe harbor provisions under the Private Securities
Litigation Reform Act of 1995. These forward-looking statements are
neither promises nor guarantees of future performance and are
subject to a variety of risks and uncertainties, many of which are
beyond our control, and may cause actual results to differ
materially from those contemplated in these forward-looking
statements. Those risks include the following: (i) we may encounter
unexpected adverse events in patients during our clinical
development of any product candidate that cause us to discontinue
further development; (ii) we may not be able to successfully
demonstrate the safety and efficacy of our product candidates at
each stage of clinical development; (iii) success in preclinical
studies or in early-stage clinical trials may not be repeated or
observed in ongoing or future studies, and ongoing or future
preclinical and clinical results may not support further
development of, or be sufficient to gain regulatory approval to
market AV-101, PH94B, and/or PH10; (iv) decisions or actions of
regulatory agencies may negatively affect the progress of, and our
ability to proceed with, further clinical studies or to obtain
marketing approval for our drug candidates; (v) we may not be able
to obtain or maintain adequate intellectual property protection and
other forms of marketing and data exclusivity for our product
candidates; (vi) we may not have access to or be able to secure
substantial additional capital to support our operations, including
our ongoing clinical development activities; and (vii) we may
encounter technical and other unexpected hurdles in the
manufacturing and development of any of our product candidates.
Certain other risks are more fully discussed in the section
entitled "Risk Factors" in our most recent annual report on Form
10-K, as well as discussions of potential risks, uncertainties, and
other important factors in our other filings with the Securities
and Exchange Commission (SEC). Our SEC filings are available on the
SEC's website at www.sec.gov.
In addition, any forward-looking statements represent our views
only as of the issuance of this release and should not be relied
upon as representing our views as of any subsequent date. We
explicitly disclaim any obligation to update any forward-looking
statements.
-2-
Company Contact
Mark A.
McPartland
VistaGen
Therapeutics Inc.
Phone:
+1 (650) 577-3600
Email: IR@vistagen.com
Investor Contact
Valter
Pinto / Allison Soss
KCSA
Strategic Communications
Phone:
+1 (212) 896-1254/+1 (212) 896-1267
Email: VistaGen@KCSA.com
Media Contact
Caitlin
Kasunich / Lisa Lipson
KCSA
Strategic Communications
Phone:
+1 (212) 896-1241/+1 (508) 843-6428
Email: VistaGen@KCSA.com
-3-
VISTAGEN THERAPEUTICS, INC.
Consolidated Balance Sheets
(Amounts in dollars, except share amounts)
|
|
June 30,
|
March 31,
|
|
|
2019
|
2019
|
|
|
(Unaudited)
|
(Note 2)
|
|
|
|
|
|
ASSETS
|
|
|
|
Current
assets:
|
|
|
|
Cash
and cash equivalents
|
$8,297,100
|
$13,100,300
|
|
Receivable
from supplier
|
-
|
300,000
|
|
Prepaid
expenses and other current assets
|
482,600
|
250,900
|
|
Total
current assets
|
8,779,700
|
13,651,200
|
|
Property
and equipment, net
|
286,500
|
312,700
|
|
Right
of use asset - operating lease
|
3,833,300
|
-
|
|
Security
deposits and other assets
|
47,800
|
47,800
|
|
Total
assets
|
$12,947,300
|
$14,011,700
|
|
|
|
|
|
LIABILITIES AND STOCKHOLDERS’ EQUITY
|
|
|
|
Current
liabilities:
|
|
|
|
Accounts
payable
|
$933,900
|
$1,055,000
|
|
Accrued
expenses
|
1,847,000
|
1,685,600
|
|
Current
notes payable
|
246,400
|
57,300
|
|
Operating
lease oligation
|
278,100
|
-
|
|
Financing
lease obligation
|
3,000
|
3,000
|
|
Total
current liabilities
|
3,308,400
|
2,800,900
|
|
|
|
|
|
Non-current
liabilities:
|
|
|
|
Accrued
dividends on Series B Preferred Stock
|
4,050,700
|
3,748,200
|
|
Deferred
rent liability
|
-
|
381,100
|
|
Operating
lease obligation
|
3,956,900
|
-
|
|
Financing
lease obligation
|
5,500
|
6,300
|
|
Total
non-current liabilities
|
8,013,100
|
4,135,600
|
|
Total
liabilities
|
11,321,500
|
6,936,500
|
|
|
|
|
|
Commitments
and contingencies
|
|
|
|
Stockholders’ equity:
|
|
|
|
Preferred
stock, $0.001 par value; 10,000,000 shares authorized at June 30,
2019 and March 31, 2019
|
|
|
|
Series
A Preferred, 500,000 shares authorized, issued and outstanding at
June 30, 2019 and March 31, 2019
|
500
|
500
|
|
Series
B Preferred; 4,000,000 shares authorized at June 30, 2019 and March
31, 2019;
|
||
|
1,160,240
shares issued and outstanding at June 30, 2019 and March 31,
2019
|
1,200
|
1,200
|
|
Series
C Preferred; 3,000,000 shares authorized at June 30, 2019 and March
31, 2019;
|
||
|
2,318,012 shares issued and outstanding at June 30, 2019 and March
31, 2019
|
2,300
|
2,300
|
|
Common
stock, $0.001 par value; 100,000,000 shares authorized at June 30,
2019 and March 31, 2019;
|
||
|
42,758,630
shares issued and outstanding at June 30, 2019 and March 31,
2019
|
42,800
|
42,800
|
|
Additional
paid-in capital
|
192,890,400
|
192,129,900
|
|
Treasury
stock, at cost, 135,665 shares of common stock held at June 30,
2019 and March 31, 2019
|
(3,968,100)
|
(3,968,100)
|
|
Accumulated
deficit
|
(187,343,300)
|
(181,133,400)
|
|
Total
stockholders’ equity
|
1,625,800
|
7,075,200
|
|
Total
liabilities and stockholders’ equity
|
$12,947,300
|
$14,011,700
|
-4-
VISTAGEN THERAPEUTICS, INC.
STATEMENTS
OF OPERATIONS
(Amounts in dollars, except share
amounts)
(Unaudited)
|
|
Three Months Ended June 30,
|
|
|
|
2019
|
2018
|
|
Operating
expenses:
|
|
|
|
Research
and development
|
$4,313,900
|
$2,743,700
|
|
General
and administrative
|
1,910,100
|
1,466,300
|
|
Total
operating expenses
|
6,224,000
|
4,210,000
|
|
Loss
from operations
|
(6,224,000)
|
(4,210,000)
|
|
Other
income (expenses), net:
|
|
|
|
Interest
income (expense), net
|
16,500
|
(2,100)
|
|
Loss
before income taxes
|
(6,207,500)
|
(4,212,100)
|
|
Income
taxes
|
(2,400)
|
(2,400)
|
|
Net
loss and comprehensive loss
|
$(6,209,900)
|
$(4,214,500)
|
|
|
|
|
|
Accrued
dividend on Series B Preferred stock
|
(302,500)
|
(273,500)
|
|
|
|
|
|
Net
loss attributable to common stockholders
|
$(6,512,400)
|
$(4,488,000)
|
|
|
|
|
|
Basic
and diluted net loss attributable to common
|
|
|
|
stockholders
per common share
|
$(0.15)
|
$(0.20)
|
|
|
|
|
|
Weighted
average shares used in computing
|
|
|
|
basic
and diluted net loss attributable to common
|
|
|
|
stockholders
per common share
|
42,622,965
|
22,987,066
|
-5-
Exhibit 99.2

VistaGen Therapeutics Achieves Target Patient Enrollment in the
ELEVATE Study of AV-101 as an Adjunctive Treatment for Major
Depressive Disorder
Company on Track to Report Topline Data Before Year
End
SOUTH SAN FRANCISCO, Calif., August 15, 2019 – VistaGen
Therapeutics (NASDAQ: VTGN), a clinical-stage
biopharmaceutical company developing new generation medicines for
central nervous system (CNS) diseases and disorders with high unmet
need, announced today that the Company has achieved completion of
target patient enrollment (n = 180) in its Phase 2 ELEVATE clinical
trial. ELEVATE is a multi-center, double-blind, placebo-controlled
clinical study to evaluate the efficacy and safety of AV-101,
VistaGen’s novel, oral NMDA (N-methyl-D-aspartate) receptor
glycine site antagonist, as an adjunctive treatment (together with
an FDA-approved oral antidepressant (AD)) for major depressive
disorder (MDD) in adult patients with an inadequate therapeutic
response to their current AD.
VistaGen expects to report topline results from the Phase 2 ELEVATE
study before the year end 2019.
“We are very encouraged to reach this important milestone in
our Phase 2 development program for AV-101 in MDD. Achieving target
patient enrollment puts us one step closer to redefining the
standard of care for a large and growing number of individuals who
are unable to reduce their symptoms of depression with their
current antidepressant alone,” said Shawn Singh, Chief
Executive Officer,
VistaGen. “We
look forward to completing the ELEVATE study in the near term and
reporting topline data by year end.”
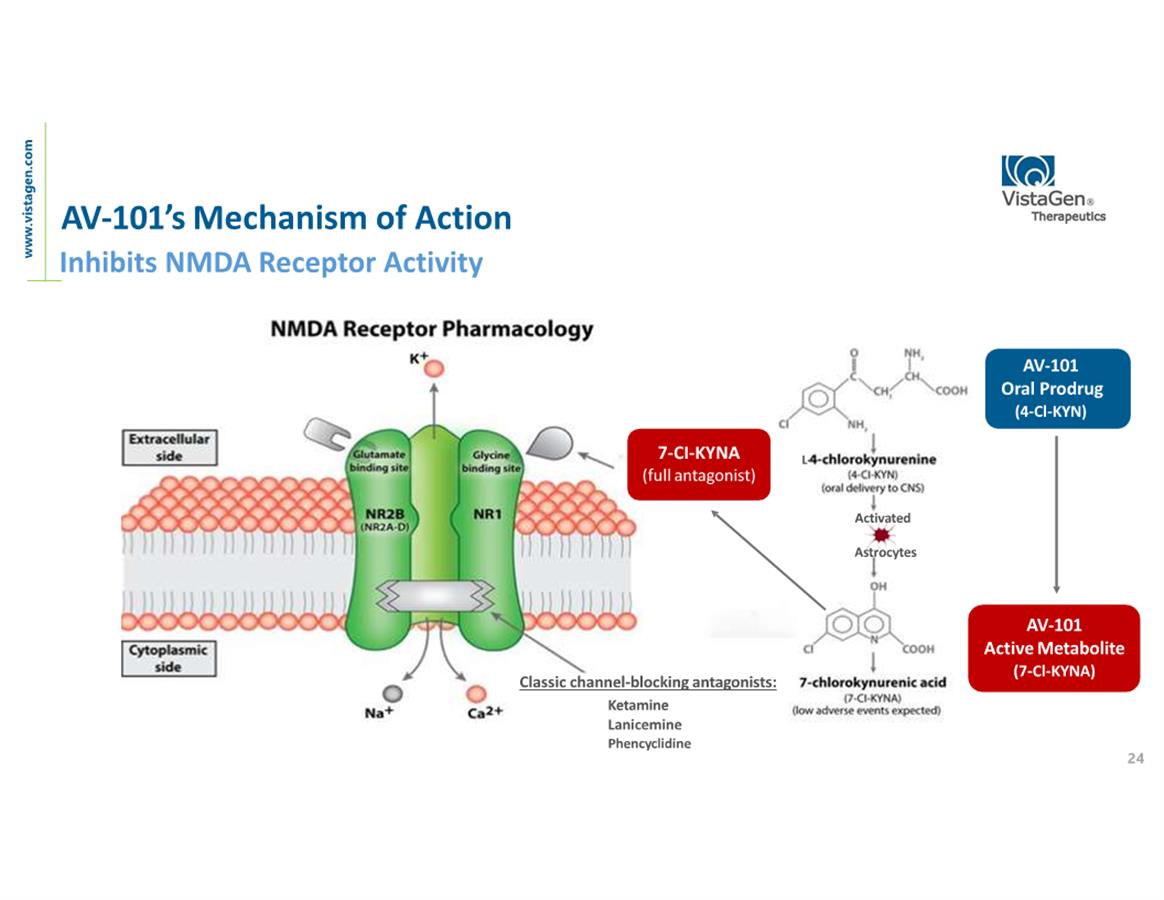
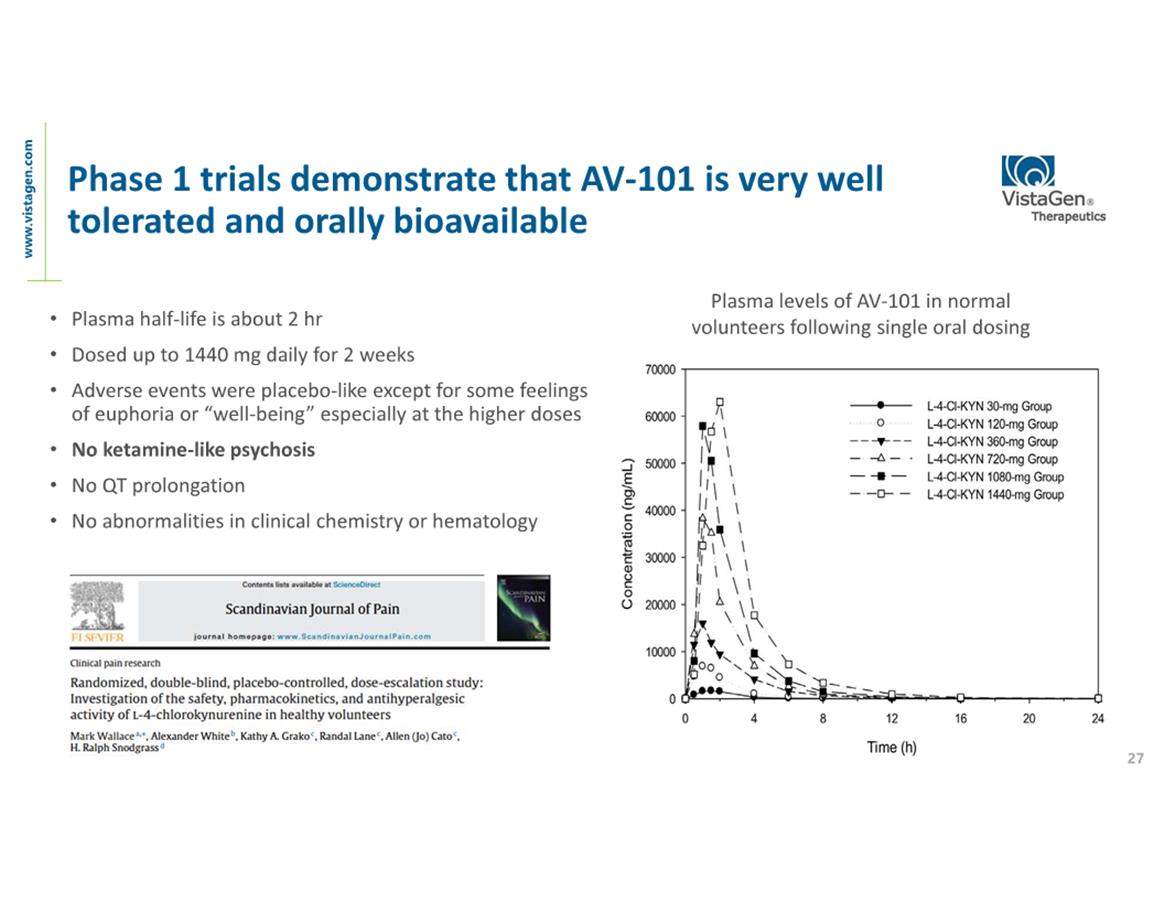
About AV-101
AV-101
(4-Cl-KYN) belongs to a new generation of investigational medicines
in neuropsychiatry and neurology known as NMDA
(N-methyl-D-aspartate) glutamate receptor modulators. The NMDA
receptor is a pivotal receptor in the brain and abnormal NMDA
function is associated with multiple CNS diseases and disorders,
including chronic neuropathic pain, epilepsy, MDD, levodopa-induced
dyskinesia (LID) and many others. AV-101 is an oral prodrug of
7-Cl-KYNA which binds uniquely at the glycine site of the NMDA
receptor. With its exceptional safety profile in all studies to
date, AV-101 has potential to be a new at-home treatment for
multiple large market CNS indications where current treatments are
inadequate to meet high unmet patient needs. VistaGen is currently
focused on potential development of AV-101 for MDD, neuropathic
pain, suicidal ideation and dyskinesia associated with levodopa
treatment for Parkinson’s disease. The FDA has granted Fast
Track designation for development of AV-101 as both a
potential adjunctive treatment
for MDD and as a non-opioid treatment
for neuropathic pain.
-1-
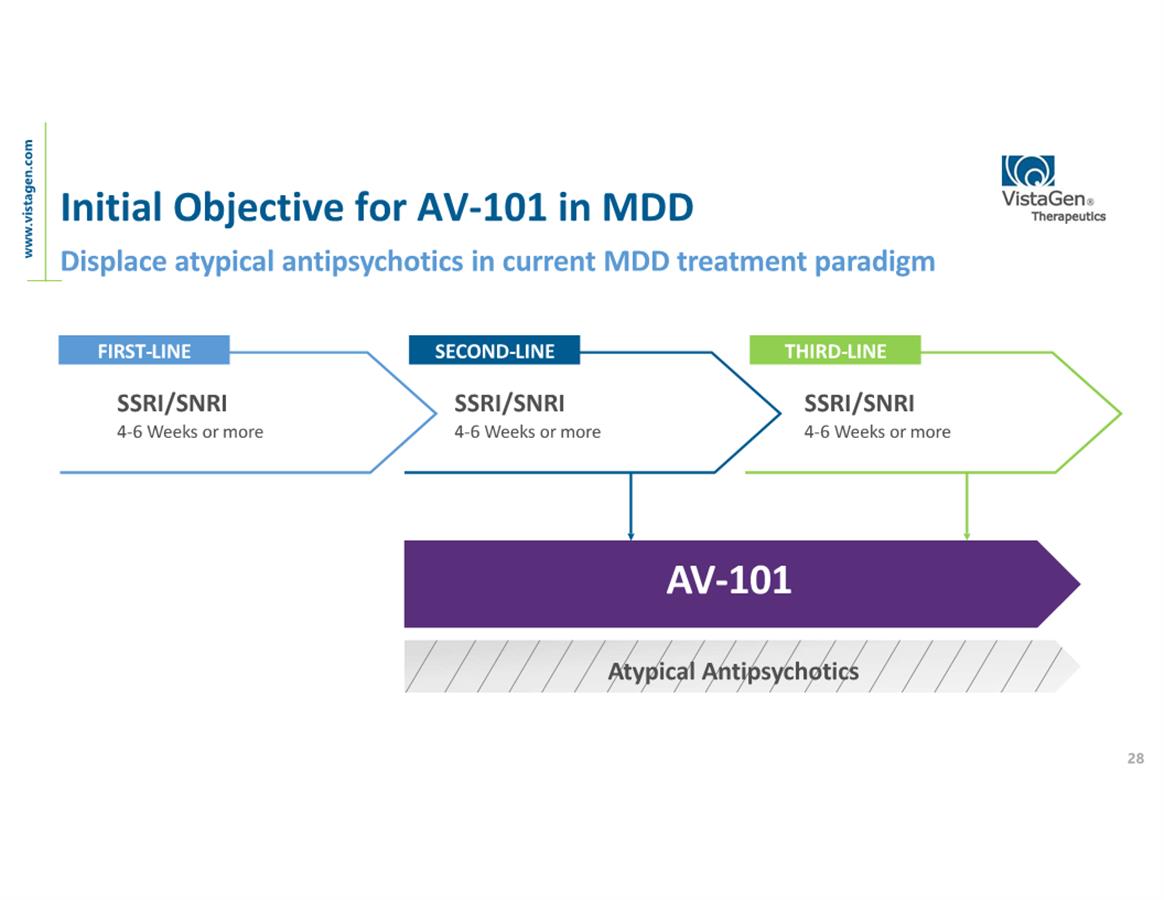
About ELEVATE
Among VistaGen's objectives for AV-101 in MDD is to replace
atypical antipsychotics in the current MDD drug treatment paradigm
and redefine the standard of care for individuals who are unable to
reduce their symptoms of depression with their current
antidepressant alone. The ELEVATE study is an ongoing U.S.
multi-center, randomized, double-blind, placebo-controlled Phase 2
clinical study to evaluate the efficacy and safety of adjunctive
use of AV-101 in adult MDD patients who have an inadequate response
to standard FDA-approved antidepressant therapy, either a selective
serotonin reuptake inhibitor (SSRI), a serotonin norepinephrine
reuptake inhibitor (SNRI), or bupropion. The primary endpoint of
the study is the change from baseline on the Montgomery-Åsberg
Depression Rating Scale (MADRS-10) total score.
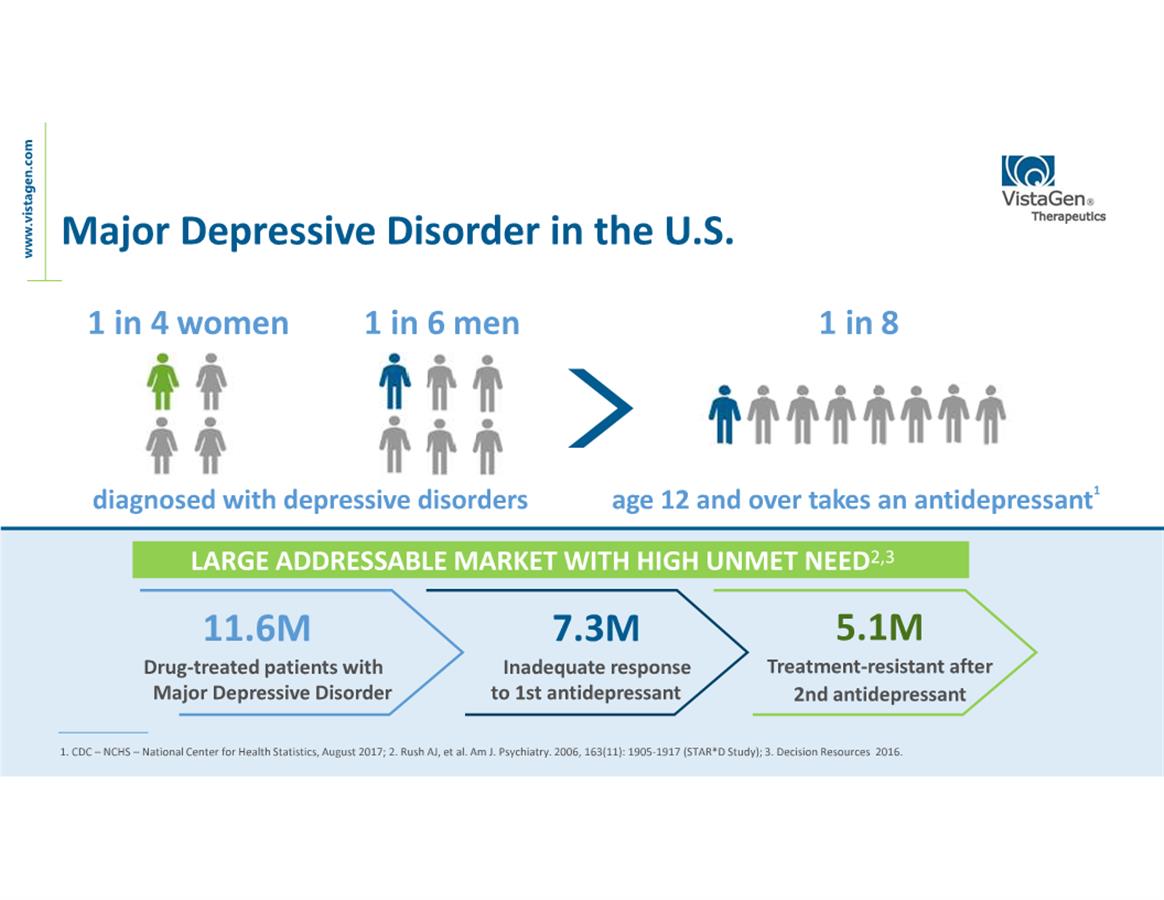
About Major Depressive Disorder (MDD)
MDD is a serious neurobiologically-based mood disorder, affecting
approximately 16 million adults in the U.S., according to the NIMH.
Individuals diagnosed with MDD exhibit depressive symptoms, such as
a depressed mood or a loss of interest or pleasure in daily
activities, for more than a two-week period, as well as impaired
social, occupational, educational or other important functioning
which has a negative impact on their quality of life. Globally, MDD
affects nearly 300 million people of all ages and is the leading
cause of disability worldwide.
About VistaGen
VistaGen
Therapeutics is a clinical-stage biopharmaceutical company
developing new generation medicines for CNS diseases and disorders
where current treatments are inadequate, resulting in high unmet
need. VistaGen's pipeline includes
three differentiated, clinical-stage CNS drug candidates, AV-101,
PH10 and PH94B, each with an exceptional safety profile in all
clinical studies to date and therapeutic potential in multiple
large and growing CNS markets. For more information, please
visit www.vistagen.com and
connect with VistaGen on Twitter, LinkedIn and Facebook.
Forward-Looking Statements
This release contains various statements concerning VistaGen's
future expectations, plans and prospects, including without
limitation, our expectations regarding development and
commercialization of our three drug candidates, (i) AV-101 for MDD,
and specifically the completion of our ELEVATE study, NP, LID and
suicidal ideation; (ii) PH94B for SAD; and (iii) PH10 for MDD. In
addition, statements concerning the Company’s future
expectations may include statements regarding intellectual property
and commercial protection of our drug candidates. Each of these
statements constitute forward-looking statements for the purposes
of the safe harbor provisions under the Private Securities
Litigation Reform Act of 1995. These forward-looking statements are
neither promises nor guarantees of future performance and are
subject to a variety of risks and uncertainties, many of which are
beyond our control, and may cause actual results to differ
materially from those contemplated in these forward-looking
statements. Among these risks is the possibility that (i) we may
encounter unexpected adverse events in patients during our clinical
development of any product candidate, including in AV-101 during
the ELEVATE study, that cause us to discontinue further
development, (ii) we may not be able to successfully demonstrate
the safety and efficacy of our product candidates at each stage of
clinical development, including for AV-101 during the ELEVATE
study, (iii) success in preclinical studies or in early-stage
clinical trials may not be repeated or observed in ongoing or
future studies, and ongoing or future preclinical and clinical
results may not support further development of, or be sufficient to
gain regulatory approval to market AV-101, (iv) decisions or
actions of regulatory agencies may negatively affect the progress
of, and our ability to proceed with, the ELEVATE study or other
further clinical studies or to obtain marketing approval for our
drug candidates, (v) we may not be able to obtain or maintain
adequate intellectual property protection and other forms of
marketing and data exclusivity for our product candidates, (vi) we
may not have access to or be able to secure substantial additional
capital to support our operations, including our ongoing clinical
development activities, and (vii) we may encounter technical and
other unexpected hurdles in the manufacturing and development of
any of our product candidates. Certain other risks are more fully
discussed in the section entitled "Risk Factors" in our most recent
annual report on Form 10-K and subsequent quarterly reports on Form
10-Q, as well as discussions of potential risks, uncertainties, and
other important factors in our other filings with the Securities
and Exchange Commission (SEC). Our SEC filings are available on the
SEC's website at www.sec.gov.
In addition, any forward-looking statements represent our views
only as of the issuance of this release and should not be relied
upon as representing our views as of any subsequent date. We
explicitly disclaim any obligation to update any forward-looking
statements.
-2-
Company Contact
Mark A.
McPartland
VistaGen
Therapeutics Inc.
Phone:
+1 (650) 577-3600
Email:
IR@vistagen.com
Investor Contact
Valter
Pinto / Allison Soss
KCSA
Strategic Communications
Phone:
+1 (212) 896-1254/+1 (212) 896-1267
Email:
VistaGen@KCSA.com
Media Contact
Caitlin
Kasunich / Lisa Lipson
KCSA
Strategic Communications
Phone:
+1 (212) 896-1241/+1 (508) 843-6428
Email:
VistaGen@KCSA.com
###
-3-
Exhibit
99.3